

Properties like atomic radius, ionic radius, ionization energy, electro-negativity, affinity and valence of electrons, etc., called atomic properties.Ītomic Radius- The size in the atom of any product from the middle of the nucleus to the electrons ' outermost shell is called its atomic radius. Periodicity: The occurrence of similar properties after regular intervals is called periodicity.Ĭause of Periodicity: The properties of the elements are that similar electronic product configuration occurs regularly as the number of atoms increases.Ītomic Properties: The physical traits of an element's atom are called atomic properties. So the outermost shell of noble gasses is filled up entirely. The general electronic outermost configuration of noble gases (except for He) is ns2 np6. Noble Gases: Group 18 gaseous elements are called noble gasses. The outer electronic configuration is characteristic of (n-2) f1-14 (n-1) d0-1 ns2. Actinoids distinguished by 5f-orbitals filling are the elements from Th 103Lr following actinium. Atomic radius usually decreases over a period of time, and increases as we descend the group.į-Block Elements (Inner- Transition Series)Ĭharacterized by the filling of 4 f-orbitals, the lanthanum-following elements are from 58Ce to 71Lu. There are four transformation sequences 3d, 4d, 5d, and 6d. He (1s2) is a s-block element but is positioned with the group 18 elements (ns2 np6) because it has a fully filled valence shell and thus exhibits characteristic properties of other noble gasses.ĭ- block Elements (Transition Elements) : They are community 3 to 12 elements having external electronic configuration(n-1) d1-10 ns1-2. The most electronic parameter outer is ns2 np1-6. P- Block Elements: They are part of group-13 to 18. S- block Elements: Group-1 (Alkali metals) and Group-2 (Alkaline earth metals) elements with outermost electronic configurations of ns1 and ns2, respectively. The elements of the s-and p-block are called major group elements, or representative elements.
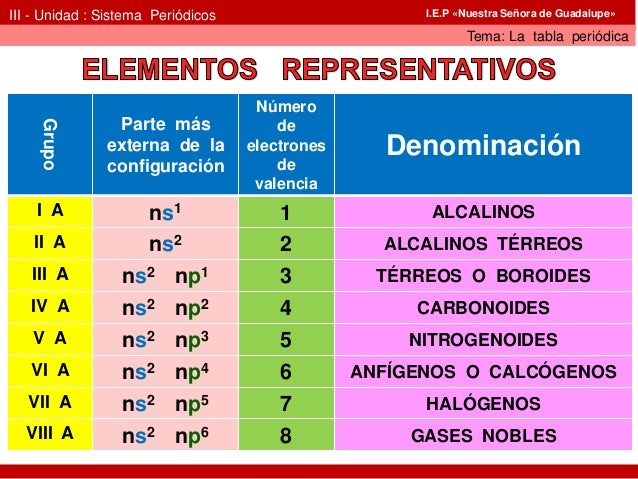
Main Group Elements/ Representative Elements: Elements are Classified into: s-, p-, d- and f- blocks. Modern Periodic Law: Elements physical and chemical properties are the periodic features of their atomic numbers. The position of an element in the periodic table therefore depends on its atomic number as compared with its atomic mass. Moseley, the English physicist has shown that an element's atomic numbers are more fundamental properties than its atomic mass. Mandeleev’s Periodic Law:- The elements ' properties are the periodic function of their atomic masses.
